Crystals and crystalline growth have been studied for centuries because of, at least in part, their symmetry. Crystals are cut in such a way as to increase this symmetry even more, because most people find symmetry attractive. However, where does the original symmetry in a crystal come from? Without it, jewelers who cut gemstones would not exist, for the symmetry of crystalline minerals themselves is what gives such professionals the raw materials with which to work.
To understand anything about how crystals grow, one must look at a bit of chemistry. The growth of crystals:
- Involves very small pieces: atoms, molecules, ions, and/or polyatomic ions
- Involves a small set of simple rules for how these small pieces attach to each other
Why small pieces? That’s easy: we live in a universe where atoms are tiny, compared to anything we can see. Why is the number of rules for combining parts small, though? Well, in some materials, there are, instead, large numbers of ways that atoms, etc., arrange themselves — and when that happens, the result, on the scale we can see, is simply a mess. Keep the number of ways parts can combine extremely limited, though, and it is more likely that the result will possess the symmetry which is the source of the aesthetic appeal of crystals.
This can be modeled, mathematically, by using polyhedral clusters. For example, I can take a tetrahedron, and them augment each of its four faces with a rhombicosidodecahedron. The result is this tetrahedral cluster:
Next, having chosen my building blocks, I need a set of rules for combining them. I choose, for this example, these three:
- Only attach one tetrahedral cluster of rhombicosidodechedra to another at triangular faces — and only use those four triangles, one on each rhombicosidodecahedron, which are at the greatest distance from the cluster’s center.
- Don’t allow one tetrahedral cluster to overlap another one.
- When you add a tetrahedral cluster in one location, also add others which are in identical locations in the overall, growing cluster.
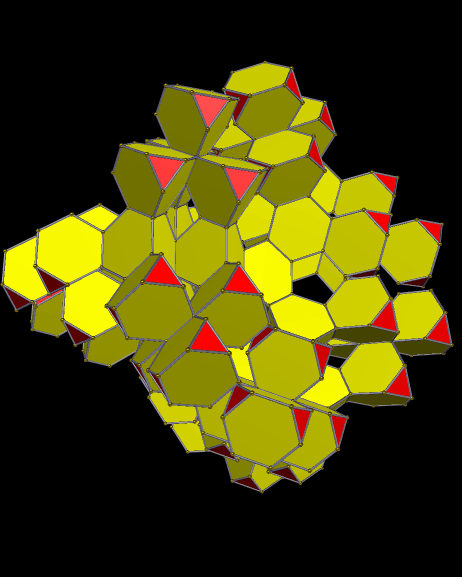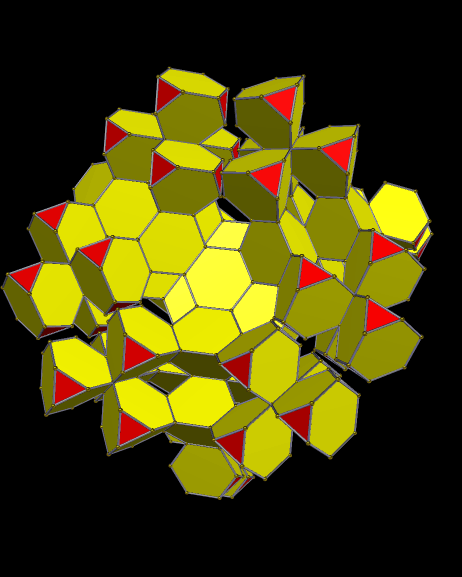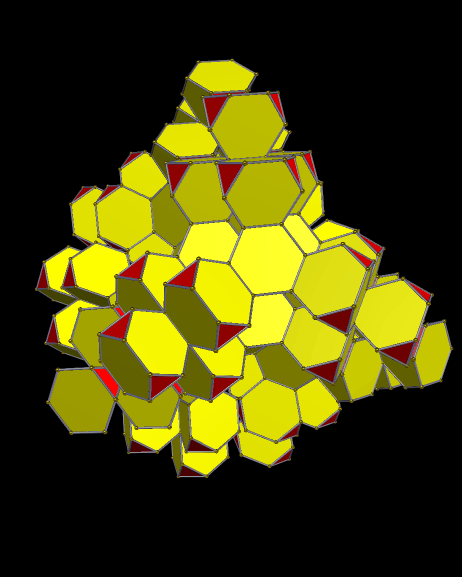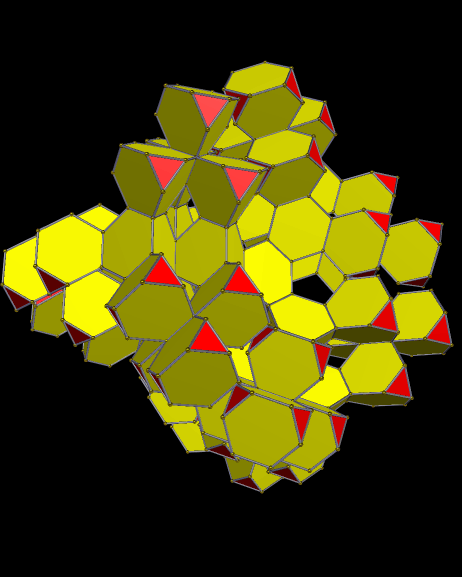
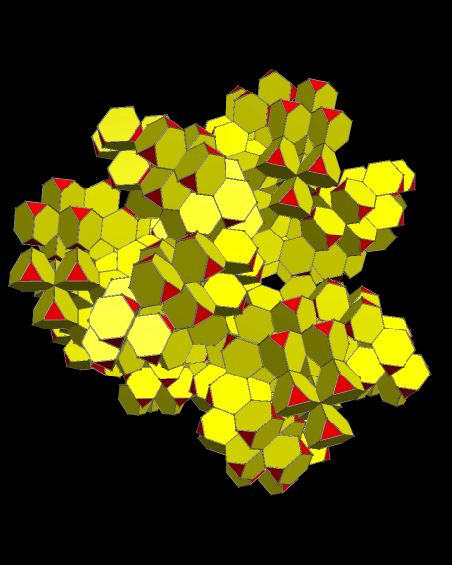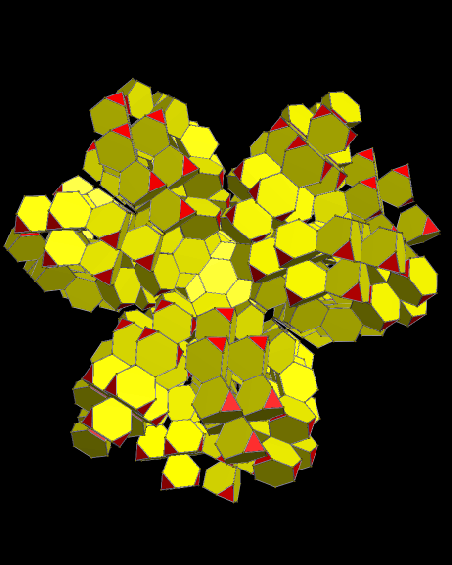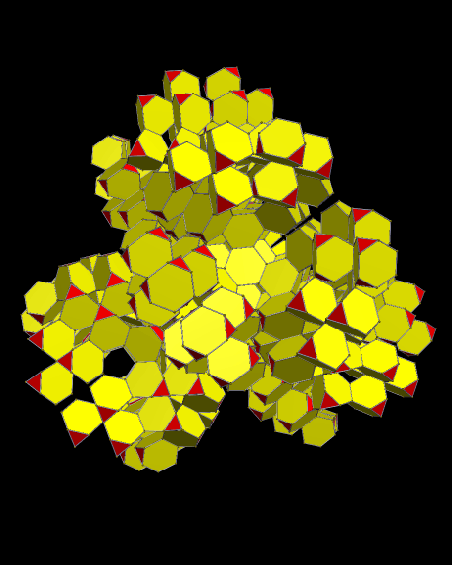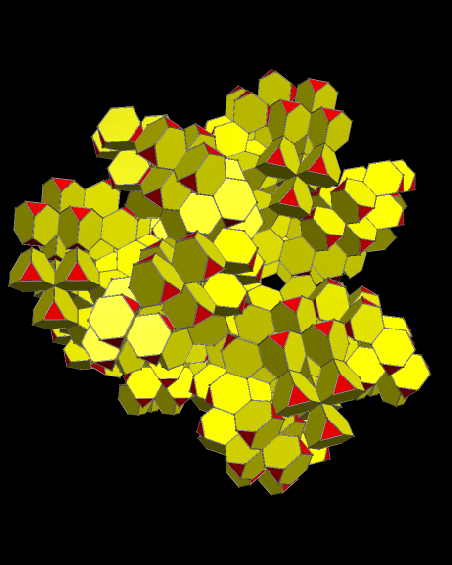
Using these rules, the first augmentation produces this:
That, in turn, leads to this:
Next, after another round of augmentation:
One more:
In nature, of course, far more steps than this are needed to produce a crystal large enough to be visible. Different crystals, of course, have different shapes and symmetries. How can this simulation-method be altered to model different types of crystalline growth? Simple: use different polyhedra, and/or change the rules you select as augmentation guidelines, and you’ll get a different result.
[Note: all of these images were created using Stella 4d: Polyhedron Navigator. This program is available at http://www.software3d.com/Stella.php.]