
I propose that 384,400 km (238,855 miles), the average distance from the Earth to the Moon, be called a “moon unit.” Example: “The mileage of my car is over one moon unit.”

I propose that 384,400 km (238,855 miles), the average distance from the Earth to the Moon, be called a “moon unit.” Example: “The mileage of my car is over one moon unit.”

Math jokes are almost universally awful — or, at least, it seems that way to me, since I spend a lot of time around ninth graders. Hearing “Gee, I’m a tree” or “Pi are square? No, pies are round, and cake are square!” will generally elicit a groan from me, and each new cadre of students seems to think they invented these fossilized puns. An even worse “joke” is the giggling one should expect from, say, 7th graders, if one squares the number thirteen in their presence.
I do know exactly one good math joke, though. I didn’t hear it from a student. If you’re curious, read on. Only the embellishments are original; I didn’t make up the joke, itself, though, nor do I know who did.
My source for the image below is this fellow WordPress blogger’s photography blog.

So a physicist, chemist, and a lawyer enter a balloon race together. Theirs is the last balloon to leave, because the lawyer had been in court, arrived late, and caused a short delay in departure. The consequences of this were serious, though, for a sudden cross-wind blew them off course, right after takeoff. Soon, they couldn’t even see any of the other balloons in the race, and none of them recognized any landmarks in the landscape below.
Soon, they had no idea where they were, and started getting worried about making it to their next classes on time — or back to court, in the case of the lawyer. The chemist was particularly worried. “What are we going to do?” asked the chemist.
The physicist replies, “I have an idea!” He cups his hands, leans out, and yells, as loud as he can, “Hello! Where are we?”
The balloon flies on for at least two long, anxious minutes as the trio waits, silently, for an answer. Eventually, they hear, from a great distance, a voice. “Hello! You’re lost!”
The physicist looks at the other two, and says, “That, my friend, was a mathematician.”
“How,” asked the lawyer, “could you possibly know that?”
“Three things,” replied the physicist. He held up one finger. “First, it took him a long time to answer.”
“Second,” he continued, holding up two fingers, “the answer, when it finally came, was absolutely correct.”
A third finger joined the first two. “Third, the answer, when it finally came, was completely useless!”
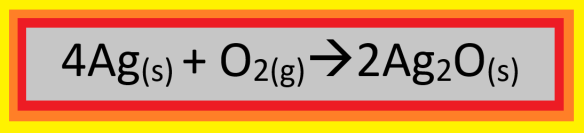
Because of the price of silver being literally on fire, they will not be buying and selling troy ounces of metallic silver when the markets open in New York tomorrow morning. Instead, they will be selling “oxide ounces” of silver oxide, in sealed-plastic capsules of this black powder, with an oxide ounce of silver oxide being defined as that amount of silver oxide which contains one troy ounce of silver.

A troy ounce of silver is 31.1 grams of that element, which has a molar mass of 107.868 g/mole. Therefore, a troy ounce of silver contains (31.1 g)(1 mol/107.868 g) = 0.288 moles of silver. An oxide ounce of silver oxide would also contain oxygen, of course, and the formula on the front side of a silver oxide capsule (shown above; information on the back of the capsule gives the number of oxide ounces, which can vary from one capsule to another) is all that is needed to know that the number of moles of oxygen atoms (not molecules) is half the number of moles of silver, or (0.288 mol)/2 = 0.144 moles of oxygen atoms. Oxygen’s non-molecular molar mass is 15.9994 g, so this is (0.144 mol)(15.9994 g/mol) = 2.30 g of oxygen. Add that to the 31.1 g of silver in an oxide ounce of silver oxide, and you have 31.1 g + 2.30 g = 33.4 grams of silver oxide in an oxide ounce of that compound.
In practice, however, silver oxide (a black powder) is much less human-friendly than metallic silver bars, coins, or rounds. As you can easily verify for yourself using Google, silver oxide powder can, and has, caused health problems in humans, especially when inhaled. This is the reason for encapsulation in plastic, and the plastic, for health reasons, must be far more substantial than a mere plastic bag. For encapsulated silver oxide, the new industry standard will be to use exactly 6.6 g of hard plastic per oxide ounce of silver oxide, and this standard will be maintained when they begin manufacturing bars, rounds, and coins of silver oxide powder enclosed in hard plastic. This has created a new unit of measure — the “encapsulated ounce” — which is the total mass of one oxide ounce of silver oxide, plus the hard plastic surrounding it on all sides, for a total of 33.4 g + 6.6 g = 40.0 grams, which will certainly be a convenient number to use, compared to its predecessor-units.
# # #
[This is not from The Onion. We promise. It is, rather, a production of the Committee to Give Up on Getting People to Ever Understand the Meaning of the Word “Literally,” or CGUGPEUMWL, which is fun to try to pronounce.]
In the Summer of 2014, with many other science teachers, I took a four-day-long A.P. Physics training session, which was definitely a valuable experience, for me, as a teacher. On the last day of this training, though, in the late afternoon, as the trainer and trainees were winding things up, some of us, including me, started getting a little silly. Physics teachers, of course, have their own version of silly behavior. Here’s what happened.
The trainer: “Let’s see how well you understand the different forces which can serve as centripetal forces, in different situations. When I twirl a ball, on a string, in a horizontal circle, what is the centripetal force?”
The class of trainees, in unison: “Tension!”
Trainer: “In the Bohr model of a hydrogen atom, the force keeping the electron traveling in a circle around the proton is the . . . ?”
Class: “Electromagnetic force!”
Trainer: “What force serves as the centripetal force keeping the Earth in orbit around the Sun?”
Me, loudly, before any of my classmates could answer: “God’s will!”
I was, remember, surrounded by physics teachers. It took the trainer several minutes to restore order, after that.
Clearly, this requires some explanation.
January 12 is my birthday, and today is July 13, 2015.
How long until my next birthday, starting at midnight, tonight?
Since the number of days between the end of my last birthday, and midnight tonight, is exactly the same as my number of pre-birthday days which follow midnight, it follows that midnight tonight is the one point in time, this year, which is as far away from my birthday as one can get, on the calendar. The fact that antibirthdays are usually points in time, rather than full days, is a consequence of the fact that most years have an odd number of days. Subtract one for my birthday (or anyone’s, except for those rare people born on February 29 — we’ll get to them later), and 364 days remain in most years. Divide this by two, and there are 182 days to fall on either side of an antibirthday midnight, for most people, during most years.
Next year, 2016, is a leap year. What will happen to my antibirthday next year, then, with its 366 days? As it turns out, next year’s antibirthday, for me, will be a full day. Why? Adding “leap day” makes it necessary to subtract two days, rather than just one, to get 364. (An even number of post-subtraction days is needed for divisibility, by two, with no remainder.) My antibirthday in 2016 will be on July 13, all day long, because there are 182 days between that day and both of my nearest birthdays — one in that antibirthday’s near past, and one in its near future.
If we don’t have the same birthday, and you want to figure out when your own antibirthday is, you can follow the pattern above, with only minor adjustments, if your birthday, like mine, falls on or before February 28. Some additional adjustments will be needed for those with birthdays in March through December, though. Why it that? Simple: my birthday occurs before February, and this isn’t true for most people. My full-day antibirthdays occur during leap years only because of this fact. If your birthday occurs after February is over, you’ll still get full-day antibirthdays every four years, but those years won’t be leap years — they’ll be one year removed from leap years, instead. Whether this means such years will immediately precede, or follow, leap years is left as an exercise for the reader.
There’s a small group of people for whom this gets even more complicated: those whose birthdays only happen every four years, on “leap day,” February 29th. Of the people I know well, only one of them, my friend Todd, was born on a leap day, and, just to be a pest, I’m going to assign him the problem of figuring out his own antibirthdays. After all, he has plenty of time for this, since the fact that he only has a birthday every four years causes him to age at 25% of the normal rate. He looks only a bit older than me, having had only a few more birthdays that I’ve had, even though he was born in 1812, and can remember the American Civil War clearly. Fortunately for him, he was still a child in the 1860s, and this saved him from actually having to fight in that war, or any other. It must be nice to have a 280-year life expectancy, Todd!
[Image credit: before turning the birthday-cake picture above upside-down, I downloaded it from this website.]

Get it?
According to this map, I live in Little, Oklahoma. I work in Rock, Louisiana, not far away. I buy most of my Chinese food (>50% of what I eat) a few kilometers to the North, in the former North Little Rock, now renamed Argenta, Missouri.
It was fun partitioning the state I live in (using the Arkansas River, and “Tornado Alley,” also known as Interstate 30, to do it), and otherwise playing around with the map of the country and continent where I live.
By the way, I actually do believe that if the USA ever falls apart, Soviet-Union-style, Texas really would be the only former state to give itself a subtitle.